Progress in the scientific understanding of water treatment kept pace with progress in engineering. The first chemical treatment for softening hard water — lime softening — was invented by the chemist Thomas Clark (like Gibb, also a Scott) in the 1830s. The process consists of adding a solution of calcium hydroxide (lime water). This process precipitates any calcium bicarbonate present in the water, along with the added calcium and some of the magnesium, as hydroxides and silicates. The precipitates settle or are filtered. Sodium carbonate (soda ash) can be added to remove the permanent hardness.
Lime softening has the added advantage of killing some microorganisms. The method was soon applied to soften water from the Thames and used in many industrial applications. It is still in use today. Warm lime softening is even more efficient, because the solubility of calcium, magnesium, and silicates decreases with temperature.
One of the driving forces of the Industrial Revolution was the steam engine invented by Thomas Newcomen and perfected by James Watt. The scaling of boilers was a major problem. It drove the development of other processes for softening water by chemical addition, such as Edward Bohlig’s hot magnesium oxide softening process, patented in the 1870s. This process was developed specifically for treating feedwater for boilers. It greatly reduced silicates, as silica adsorbs on magnesium hydroxide precipitate.
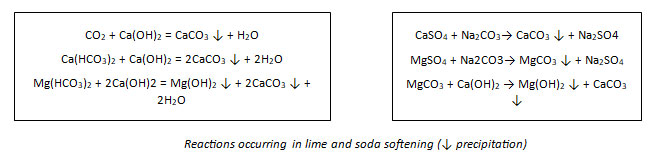
The other major discovery in the field of water softening was ion exchange. The phenomenon was discovered very early in the 1850s by Harry Stephen Meysey Thompson and John Thomas Way. They discovered that a solution of ammonium sulfate was transformed to calcium sulfate as it percolated through some types of soils. The soil was exchanging calcium for ammonium ions. These first ion-exchange materials were zeolites, a type of very porous aluminosilicate mineral. Natural zeolites were developed by the German chemist Robert Gans and were already in use in the first years of the 20th century, but they did not have a large capacity for removing ions. Their commercial success and widespread use was made possible when Gans invented and patented synthetic zeolites. The Permutit Company developed both industrial and domestic ion exchangers, which were widely advertised and sold.
The development of phenolic-formaldehyde resin ion exchangers was the next step, soon followed by the development of styrene-divinylbenzene polymer matrices that have not been superseded to this day. The improvements made possible by these resins did not just involve a large increase in the capacity of the materials, but also a greatly reduced regeneration time.
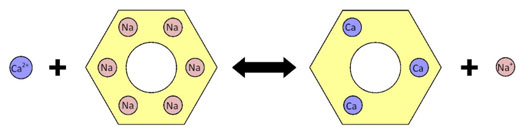
Improvements in resin ion exchangers increased capacity and reduced regeneration time.
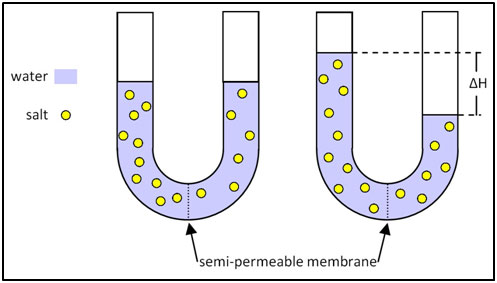
The latest widely used technology to remove hardness from water is reverse osmosis, although the phenomenon of osmosis had been known since the mid-18th century observations of the French physicist Jean Antoine Nollet, known as the abbé Nollet. His experiments using a pig’s bladder showed that a solvent would flow through a membrane from a low to a high concentration of solute, even as the membrane was impermeable to the solute. This flow would create a pressure head on the side of high solute concentration. In theory, it would thus be possible to purify water by pressurizing it on a membrane through which ions cannot pass. Research began in the 1940s, with the aim of creating a desalination technology that would be cheaper than distillation. However, it was not until 1959 that UCLA researcher Samuel Yuster and his students, Sidney Loeb and Srinivasa Sourirajan, succeeded in creating a useable system. Their membrane was made of cast cellulose acetate, but other materials were soon developed, although they involved sophisticated polymerization technologies. Within a few years, reverse osmosis plants were established that produced millions of gallons of desalinated water. The technology is also used to provide feedwater to boilers, reducing scaling while allowing high cycles of concentration. High capital cost and membrane fouling and deterioration are the major problems of these systems, though improvements continue to be made on both fronts.
 |
|
Find a Water Treater Near You: Connect with a company in your area.